Modern approaches to the correction of hemodynamic disorders in patients with severe combined trauma
Background. Selection of a rational program of infusion therapy in severe trauma remains a current problem. Crystalloid solutions are used as the principal infusion media, but literature on the effects of balanced multicomponent solutions on hemodynamic profiles in patients with severe trauma is virtually absent.
Objective: to investigate the possibilities for correction of hemodynamic disturbances in patients with severe combined trauma using a modern multicomponent product based on a 6% solution of sorbitol (Rheosorbilact).
Material and methods: The investigators have evaluated a total of 60 patients with severe combined trauma and clinical presentation of I–II degree traumatic shock, aged 18 to 46 years. The patients were divided into two groups: Group 1 (n = 30) included the patients where a standard regimen of intensive care (IС) was supplemented with Rheosorbilact administered at the dose of 8–10 ml/kg body weight; and Group 2 (n = 30), which included the patients who received a standard IС regimen.
Results: Administration of Rheosorbilact produced a reduction in extracellular fluid volume, an increase in circulating blood volume (CBV) and a transition from hypokinetic circulation to eukinetic circulation. This effect was evident already during the first hours after the start of the infusion and persisted up to 6 hours after the end of the infusion (p <0.01). The use of adrenomimetics was required before the start of the study in 7 patients (23.3%) and in 6 patients (20.0%) in Group 1 and Group 2, respectively; in 6 hours, it was required in 1 (3.3%) and in 4 (13.3%) patients, respectively. In 24 hours after administration of Rheosorbilact, none of the patients in the first group needed adrenomimetics as opposed to 3 such patients (10.0 %) in Group 2 (р <0.05).
Conclusions: The use of balanced multicomponent sorbitol-based solutions is accompanied by a change from hypokinetic circulation to eukinetic circulation, absence of interstitial hyperhydration, rapid stabilization of hemodynamic parameters and reduced need for adrenergic support.
Conflicting interests: none.
Key words: severe trauma, infusion therapy, Rheosorbilact.
Selection of a rational program of infusion therapy in severe trauma remains a current problem. Almost all shock condition require large infusion volumes due to serious depletion of intravascular volume. The concept of primary fluid resuscitation of critically injured patients is based on the strategy of permissive (controlled) hypotension as part of the damage control approach [1]. Infusion therapy should not regard achievement of normotension as its main objective, but rather focus on the following criteria:
• maintaining the patient’s consciousness;
• in penetrating wounds (without a concomitant severe traumatic brain injury): maintaining systolic blood pressure at the level of 70-80 mm Hg and at 90 mm Hg in a blunt trauma;
• in a concomitant traumatic brain injury: maintaining mean blood pressure at not less than 65 mm Hg. In case of combined trauma, the priority is to protect the brain, that is, the so-called TTB approach (Time To Brain). In this respect, an adequate and rationally structured infusion therapy is becoming very important [2].
In treatment of patients with severe trauma, the rationale for using infusion therapy is to replenish hypovolemia, correct the disorders of fluid-electrolyte and acid-base balances, improve blood rheology and disorders of microcirculation and metabolism, with the goal of effective oxygen transportation and detoxification. Infusion therapy has a multifaceted effect on the fundamental life support systems in the body. The distinctive principal effects include volemic, rheological and hemodilution effects [3].
Crystalloid and colloid solutions are used as basic infusion media. In terms of composition, isotonic electrolyte solutions approximate the extracellular fluid; however, they have a modest direct volemic effect (not more than 20% of the administered volume of the medium) [4]. Among the colloid solutions, hydroxyethyl starch solutions are getting increasingly popular in the last years due to their powerful volemic effect and prolonged elimination half-lives with a relatively small number of adverse reactions [4].
What solutions are recommended for correction of hemodynamic disorders in modern guidelines? According to the Guidelines on initial resuscitation of patients with severe combined trauma (NICE guidelines, 2016. Major trauma: Assessment and Initial Management), crystalloids are believed to be the starter infusion therapy agents for the injured patients (grade 1B recommendation). The use of hypertonic saline solutions should be considered in patients with unstable hemodynamics (grade 2B recommendation). The use of colloids is possible in patients with unstable hemodynamics in the recommended limited doses (grade 2C recommendation) [5]. It should also be kept in mind that the Damage Control principles should be routinely applied in critically injured patients with severe hemorrhagic shock, signs of ongoing bleeding and coagulopathy, hypothermia and acidosis [5].
One of the promising ways to manage impaired volemic status includes the so-called “small volume” infusions using a hyperosmolar polyelectrolyte balanced solution available as Rheosorbilact (a product of Yuria-Pharm, Ukraine). The benefits of using this solution as a first-line therapy in hypovolemic shock are primarily attributable to hyperosmolarity of this solution. The high osmolarity of the solution is created by the osmotically active substances, such as sorbitol, sodium lactate and all of the available ions, primarily the sodium ions. The osmolarity of Rheosorbilact is 891 mOsmol/kg, which is 3 times the osmolarity of plasma. The hyperosmolar effect is implemented through the following mechanisms:
• an increase in plasma osmolarity and activation of osmoreceptors, activation of the sympathetic nervous system and an increase in production of the adrenocorticotropic hormone and, as a result, epinephrine and norepinephrine;
• a reduction in capillary leak, and, respectively, reduction of fluid shift into the interstitial space;
• recruitment of additional fluid into the circulation from the interstitial space and the intracellular space according to the osmotic gradient.
An important contribution to the high osmolarity of Rheosorbilact is made by the sodium ions, whose concentration in Rheosorbilact is almost 2 times that in the blood. In fact, Rheosorbilact® contains a hypertonic saline solution. At the same time, the use of hypertonic solution of sodium chloride is known to lead to an increase in CBV and to reduction in the total volume of infusion, to improve renal function and to reduce the counts of perioperative complications and mortality in various patient populations [6, 7].
Due to its high osmolarity, Rheosorbilact causes extracellular fluid to shift into the circulation, which contributes to improvements of microcirculation and tissue perfusion [8].
The second important property is the presence of sodium lactate, a donor of alkaline reserve, which has a neutral pH and dissociates into sodium ions and lactic acid in the circulation. In the liver, the lactic acid is metabolized to sodium bicarbonate, which increases the alkalinity of blood. Unlike sodium bicarbonate, the correction of metabolic acidosis occurs in a slow fashion and without sharp fluctuations of pH values.
Of particular note is the fact that sodium lactate has been included to the WHO Model List of Essential Medicines. This WHO list consists of the most effective and safe medicinal products required to meet the most important needs of national health care systems. In the latest, the 21st Edition (2019), Sodium lactate is listed under Section 26 “Solutions correcting water, electrolyte and acid-base disturbances”, subsection 26.2 “Parenteral” [9]. Such recognition by the WHO is another confirmation of efficacy and safety of sodium lactate as a component of multifunctional intravenous solutions.
Therefore, the benefits of using Rheosorbilact as part of infusion therapy of volemic disturbances appear to be quite obvious. At the same time, the literature on its influence on hemodynamic profiles in patients with severe trauma is virtually absent. This was the rationale for the objective of this study.
Objective
The authors have undertaken to investigate the possibilities for correction of hemodynamic disturbances in patients with severe combined trauma using a modern multicomponent product based on a 6% solution of sorbitol (Rheosorbilact).
Material and methods
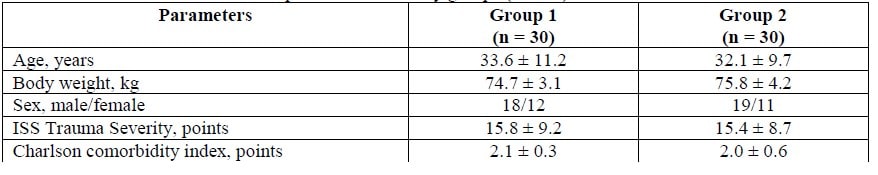
We have assessed 60 patients aged from 18 to 46 years, treated at a general type intensive care unit of Kyiv Regional Clinical Hospital. The patients were divided into two groups: Group 1 included patients with severe combined trauma and clinical presentation of I–II degree traumatic shock (n = 30), where a standard regimen of intensive care (IС) was supplemented with Rheosorbilact administered at the dose of 8–10 ml/kg body weight; Group 2 included patients with combined trauma and clinical presentation of I–II degree traumatic shock (n = 30), who received a standard IС regimen. The above mentioned groups were representative in terms of the main clinical characteristics (see Table 1).
The mean dose of Rheosorbilact in ICU patients was 8-10 ml/kg per day (600-800 ml/day). Given that Rheosorbilact is a hyperosmolar solution (with an osmolarity of approximately 900 mOsmol/L), its administration was guided by assessments of blood osmolarity, i. e. the solution was not administered in patients with confirmed or suspected hyperosmolar condition.
The efficacy of infusion therapy was evaluated based on the generally accepted clinical signs, such as systolic blood pressure (sBP), diastolic blood pressure (dBP) and mean blood pressure (mBP), heart rate (HR) and SрO2 (%), which were determined using an UM-300-10 patient monitor (manufactured by UTAS, Ukraine). After central venous line placement, central venous pressure (CVP) was measured.
Table 1. General characterization of patients across study groups (M ± m)
The changes in stroke volume index (SVI), cardiac index (CI), left ventricular power output (LVPO), ejection fraction (EF) and peripheral vascular resistance (PVR) with time were measured with echocardiography (using a SA 600 unit manufactured by Medison, The Republic of Korea). Extracellular fluid volume (EFV) was measured with a M.I. Tyshchenko’s method of integral whole body rheography using Diamant-R, a computer-assisted rheoanalyzer.
The assessments were performed at 5 time points: pre-dose and 3, 6, 12 and 24 hours after the dose of the study drug.
A conventional IС regimen was used in all patients in both study groups, which included respiratory support, analgesia and sedation, as well as elimination of disturbances of fluid and electrolyte balance and acid-base balance. The program of infusion and transfusion therapy included administration of packed red blood cells and fresh frozen plasma as indicated. Vasopressor and inotropic support was used as required. Symptomatic therapy was used.
Statistical processing of study results was performed using Microsoft Excel 2010 and Statistica 6.0 software packages. The difference between the parameters was considered to be statistically significant if the null hypothesis was correct with a likelihood of less than 5% (р <0.05).
Results and discussion
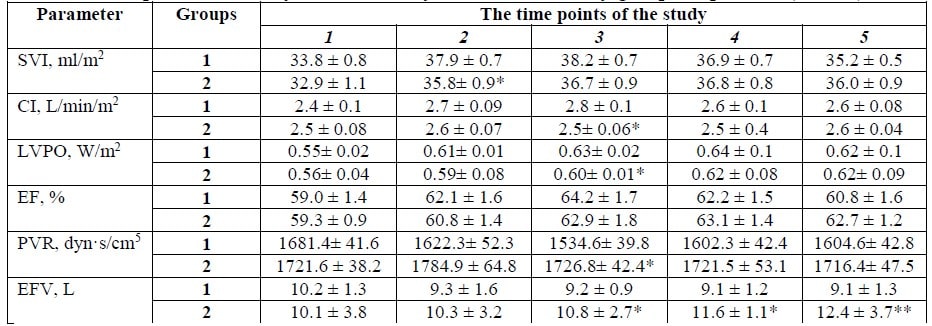
Before the start of infusion therapy, the patients in both groups had signs of moderate hypovolemia (reduced mBP values, tachycardia and decreased urine output), while imaging findings demonstrated a hypokinetic type of central hemodynamics without any significant differences between the groups of patients (see Table 2).
At the second time point in the study (3 hours after the start of infusion), there were significant increases in SVI, CI and EF by 10.4%, 12.5% and 4.8%, respectively) in the first group of patients; this being said, there was a 8.8% reduction in EFV (р <0.05) and PVR tended to decrease (a 5.3% reduction in PVR was observed). No significant changes in hemodynamic parameters have been observed in patients of the second group at this time point.
At 6 hours after the start of infusion, (time point III), a similar hemodynamic presentation persisted in Group 1: SVI, CI, LVPO and EF were significantly higher than baseline values (by 13.0%, 16.6%, 14.5% and 8.8%, respectively); PVR and EFV were decreased by 9.2% and 9.8%, respectively (р <0.05). In 12 hours after the end of the infusion (time point IV), a further increase in LVPO was noted (by 2.1% compared to the previous time point); this being said, EFV was 4.7% lower than that at the previous time point, and PVR was higher by 14.7%, respectively (р <0.05). No significant changes at these time points were observed in the second group.
Table 2. The parameters of systemic hemodynamics in the study groups of patients (M ± m)
Notes. * = the inter-group difference is significant (p < 0.05); ** = the inter-group difference is significant (p < 0.01).
By the end of the 24-hour period (time point V), the main hemodynamic parameters in both groups of patients approximated the reference values; however, in Group 1, the values of PVR and EFV were lower than the respective values in Group 2: by 6.7% and 26.6%, respectively (р <0.01).
Therefore, administration of Rheosorbilact (at 8–10 ml/kg) was leading to a significant reduction in extracellular fluid volume (via its shift into the intravascular compartment), which, along with the volume of the infused solution, was leading to an increase in CBV and to a transition from hypokinetic circulation to a more favorable eukinetic circulation. The initial manifestations of this positive hemodynamic appeared already in the first hours from the start of the infusion and persisted up to 6 hours after the end of the infusion. Subsequently, stabilization of hemodynamic parameters was observed in both groups; however, interstitial fluid volume, as at the previous time points, was significantly lower in Group 1 (p <0.01).
Analysis of clinical hemodynamic parameters has also shown interesting results (Table 3).
In course of the study, HR was found to decrease in the first group by 6.7 % at the second time point in the study and by 14.6 % in 24 hours after Rheosorbilact infusion. Conversely, mBP was 8.5% higher at the second time point in the study, which suggested an effective replenishment of CBV deficiency. Such a temporal pattern of changes was also reflected by the increasing CVP, which already in 6 hours was 112.2% higher compared to baseline values in patients of the first group, as opposed to Group 2, where CVP increased only by 17.0% against the backdrop of the infusion therapy (Table 3).
Another logical consequence of improvements in hemodynamic parameters was the lower requirement in adrenergic support in patients of the Rheosorbilact group. Thus, while before the start of the study there were 7 (23.3%) and 6 (20.0%) such patients in Group 1 and in Group 2, respectively,, already in 6 hours there were 1 (3.3 %) and 4 (13.3 %) such patients in Group 1 and in Group 2, respectively. In 24 hours after administration of Rheosorbilact, none of the patients required administration of adrenomimetics, as opposed to Group 2, where 3 patients required adrenomimetics (10.0%, p <0.05).
It should be noted that the above described IС program was generally tolerated well by the patients. In course of treatment, we have not documented any cases of crystalloid-specific adverse effects (swelling of extremities, acute heart failure) or colloid-specific adverse effects (hypocoagulation, renal impairment), and electrolyte deficiencies were quickly (within 2–3 days) replenished to normal values.
Table 3. Clinical parameters of systemic hemodynamic and adrenomimetic requirements, (M ± m)
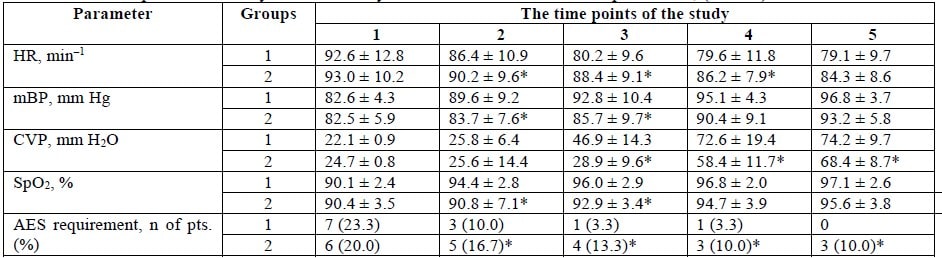
Notes. * = the difference between Group 1 and Group 2 is significant (p <0.05); AES = adrenergic support.
Conclusions
- The treatment of patients with severe combined trauma remains a current problem; solving this problem requires the use of a modern infusion therapy with balanced multicomponent solutions.
- The use of balanced multicomponent sorbitol-based solutions is accompanied by a change from hypokinetic circulation to eukinetic circulation, by absence of interstitial hyperhydration, by rapid stabilization of hemodynamic parameters and by a reduced need for adrenergic support.
- The therapeutic use of sorbitol-based solutions is well tolerated and is not accompanied by development of the complications typical for the use of either colloid solutions or classic crystalloids.
Authors:
• Oleg Anatolievich Loskutov, MD, Ph.D., Dr. Med. Sci., Professor, Head of Department of Anesthesia and Intensive Care of Shupyk National Medical Academy of Postgraduate Education
• Sergei Mikhailovich Nedashkovskyi, M.D., Ph.D., Associate Professor at the Department of Anesthesia and Intensive Care of Shupyk National Medical Academy of Postgraduate Education
• Sergei Ivanovich Babak, MD, Head of Intensive Care Unit, Kyiv Regional Clinical Hospital
• Sergei Petrovich Diachenko, MD, Physician at Intensive Care Unit, Kyiv Regional Clinical Hospital
• Boris Ivanovich Kedio, MD, Physician at Intensive Care Unit, Kyiv Regional Clinical Hospital
• Vladimir Vladimirovich Yurkiv, MD, Physician at Intensive Care Unit, Kyiv Regional Clinical Hospital
• Alexander Anatolievich Galushko, MD, Ph.D., Dr. Med. Sci., Professor at the Department of Anesthesia and Intensive Care of Shupyk National Medical Academy of Postgraduate Education
Literature:
- Cannon JW, Khan MA, Raja AS, Cohen MJ, Como JJ, Cotton BA, et al. Damage control resuscitation in patients with severe traumatic hemorrhage. Journal of Trauma and Acute Care Surgery [Internet]. Ovid Technologies (Wolters Kluwer Health); 2017 Mar;82(3):605–17. Available from: https://doi.org/10.1097/ta.0000000000001333
- Babak SI, Kuchinskaya IA, Galushko OA, Dyachenko SP. Modern management strategies for infusion therapy for severe trauma. Emergency medicine. 2016;5(4):560–7.
- Shlapak IP, Golubovska OA, Halushko OA. [Dehydration syndrome]. Acute conditions and emergencies in medical practice. 2015;6:15–9. [In Ukrainian]
- Marino Paul L. Intensive care. Per. from English under the general. ed. A.P. Zilber. M.: GEOTAR-Media, 2010;768.
- Glen J, Constanti M, Brohi K. Assessment and initial management of major trauma: summary of NICE guidance. BMJ [Internet]. BMJ; 2016 Jun 22; i3051. Available from: https://doi.org/10.1136/bmj.i3051
- Orbegozo D, Vincent JL, Creteur J, Su F. Hypertonic Saline in Human Sepsis. Anesthesia & Analgesia [Internet]. Ovid Technologies (Wolters Kluwer Health); 2019 Jun;128(6):1175–84. Available from: https://doi.org/10.1213/ane.0000000000003955
- Pfortmueller CA, Schefold JC. Hypertonic saline in critical illness – A systematic review. Journal of Critical Care [Internet]. Elsevier BV; 2017 Dec;42:168–77. Available from: https://doi.org/10.1016/j.jcrc.2017.06.019
- Cherniy VI., Kolesnikov AN., Oleinikov KN., Egorov AA., Biloshapka VI. Rational infusion therapy. Donetsk: Publisher Zaslavsky A.Yu., 2012;184. [In Ukrainian]
- 21st World Health Organization Model List of Essential Medicines 2019. Access: https://apps.who.int/iris/bitstream/handle/10665/325771/WHOMVP-EMP-IAU-2019.06-eng.pdf?ua=1