Rheosorbilact: detoxifying properties and effects of this drug on hemostasis
Summary. The administration of Rheosorbilact in the form of intravenous infusions in the setting of combined therapy of patients with purulent-septic diseases, isolated craniocerebral injuries, pneumonias, and surgical interventions for the internal carotid artery reconstruction exerts a pronounced detoxification effect, which manifests itself in the reduction in concentration of middle molecules and lipid peroxidation products. Indices of the blood coagulation system were quickly returning to normal values, thus indicating an improvement in blood rheology.
Key words: detoxification, rheology, Rheosorbilact, middle molecules, malondialdehyde.
Infusion-detoxification therapy (IDT) is a cornerstone for achieving favorable disease outcomes in the clinical practice of an anesthesiologist/resuscitator. The advancement of surgery, in particular the so-called “major surgery”, would be impossible without upgrading the support therapy, in which IDT plays a dominant role. Of course, the doctors have no doubts about the necessity for IDT, but the question “What to use?” is yet to be answered.
What are the key criteria for an anesthesiologist/resuscitator when choosing a drug product? The main aspects of infusion therapy are:
• volume correction – restoration of an adequate circulating blood volume (CBV) and normalization of its composition in case of hypovolemia;
• haemorheocorrection – normalization of homeostatic and rheological properties of blood;
• infusion rehydration – maintenance of normal micro- and macrocirculation (in particular, in clinically well-marked dehydration), normalization of electrolyte balance and acid-base balance [9].
It is common knowledge that the main disadvantage of crystalloids is their rapid redistribution from the vascular bed into the intercellular space: 75-80% of the administered drug is detected in the interstitium in 1-2 hours after the infusion. Due to such a short volemic effect, with single use of saline solutions, a larger volume of solutions is required to maintain the CBV, which entails risks of edema syndrome development [8].
Blood components include fresh frozen plasma (FFP) and albumin solution. Transfusion of FFP in clinical practice is carried out only for the prevention or correction of hemostatic disorders associated with the deficiency of blood coagulation factors. Indications for albumin transfusion can be narrowed down to the need in correction of total protein deficiency and hypoalbuminemia. Moreover, the use of albumin and FFP is limited due to their high price, which is an order of magnitude greater than the price of modern synthetic colloidal plasma substitutes [10].
Gelatin-based solutions are the least toxic products and can be used in patients with kidney diseases. Gelatin derivatives increase the plasma volume by 100%, but this effect is evanescent and lasts only for 3-4 hours [3].
When using solutions of hydroxyethyl starch, no increase in the histamine level, which occurs with the direct administration of gelatin solutions, was observed, no antigen-antibody reactions characteristic of dextran solutions were registered. However, recent data have demonstrated the negative effect of these drugs on the hemostasis system and high risk of kidney damage. Besides, high price of these drugs should be taken into account as well [11]. New prospects for detoxification therapy emerged after the development of original infusion products based on polyhydric alcohols, sodium lactate and electrolytes. One of these products is Rheosorbilact (Yuria-Pharm LLC, Ukraine). This drug exerts a multifunctional action, possesses the properties of crystalloids and colloids, corrects several types of homeostasis disorders.
Rheosorbilact contains sorbitol, basic cations (Na+, K+, Ca2+, Mg2+), anion Cl–, and lactate anion [7]. Owing to its hyperosmolarity, Rheosorbilact provokes the inflow of fluid from the intercellular space into the vascular bed, thus improving microcirculation and tissue perfusion [5]. The movement of the fluid into the intravascular bed is accompanied by an increase in the CBV. Owing to the powerful specific osmodiuretic effect of sorbitol, associated with the absence of natural mechanisms for the reabsorption of polyhydric alcohols in the proximal renal tubules in humans, the product exhibits a pronounced diuretic effect [4].
In addition, sorbitol, partially metabolized to fructose, contributes to the normalization of carbohydrate and energy metabolism, and this normalization is especially beneficial for the improvement of functional state of hepatocytes, in which the glycogen depot is restored.
The pathways of metabolism and utilization of sorbitol, fructose, and glucose are largely similar, but sorbitol has a more marked antiketogenic property. It stimulates the oxidation of fatty acids along a non-ketogenic metabolic pathway and facilitates the inclusion of ketone bodies in the Krebs cycle, thus confirming the significance of sorbitol as an energy substrate [1]. Sorbitol enhances intestinal motility due to a direct action on the neuroreceptor system of the intestinal wall and acceleration of the synthesis and secretion of villikinin, cholecystokinin and B vitamins [6].
The lactate anion contained in the drug contributes to the correction of the plasma acid-base balance, restores and stimulates the functions of the cells of the reticuloendothelial system of the liver and kidneys. All of the above effects determine the range of pharmacological properties of Rheosorbilact: anti-shock, energetic, detoxifying, diuretic actions, intestinal motility stimulation, correction of metabolic acidosis, correction of fluid-and-electrolyte balance [2].
Purpose of the study: analysis of the detoxification potential of drug product Rheosorbilact manufactured by pharmaceutical company “Yuria-Pharm LLC” (Ukraine) and its effect on the coagulation system.
Material and Methods
We examined 50 patients with purulent-septic diseases, isolated craniocerebral injuries (CCI), pneumonias, and surgical interventions for the internal carotid artery (ICA) reconstruction (Table 1). We were studying the detoxifying effect of Rheosorbilact in various toxic conditions caused by infection, injury, hypoxia and hypercapnia, that’s why the studied population is heterogenic. These patients received treatment in the departments of surgical intensive care unit No. 1 and the Second Clinic of Tashkent Medical Academy from April to June 2013. In all patients, the detoxifying action of Rheosorbilact and its effect on the coagulation system were assessed.
To assess the severity of intoxication in all patients, we used a rapid test method for determination of middle molecule (MM) concentration in blood and urine. Vast majority of authors consider MM to exert a common toxic effect as a result of their pronounced membranotropic action (Gabrielyan, N.I. et al., 1985; Galaktionov, S.G. et al., 1984; Ogai, V.Ch., 1987).
Normal MM indices in the blood are: with a spectrum of 254 nm – 0.16-0.18 nmol, with a spectrum of 286 nm – 0.126-0.136 nmol.
Normal MM indices in the urine are: with a spectrum of 254 nm – 0.5-0.9 nmol, with a spectrum of 286 nm – 0.26-0.43 nmol.
For the same purpose, we tested malondialdehyde (MDA) blood levels. MDA is formed during lipid peroxidation (LPO) and is a by-product of LPO. Accumulation of LPO products, in particular MDA, was confirmed in hypoxia conditions and certain pathological conditions accompanied by tissue hypoxia (Osinskaya, L.F., 1980; Morozov, A.V. et al., 1986).
The calculation was carried out according to the method of Andreeva L.I. et al. (1988). Normal MDA blood index is 2-3 nmol/ml.
Normal MDA urine index is 6-9 nmol/ml. The target of the infusion preparation was to improve peripheral blood circulation and remove toxins from tissue depots. This can be viewed as a process of toxin mobilization.
We set the objective to achieve the most efficient toxin mobilization from tissue depots into the central bloodstream and tested MM and MDA levels in venous blood and urine. For higher reliability, the tests were conducted at different spectrophotometer wavelengths: 254 nm and 286 nm.
Table 1. Breakdown of patients by ICD codes
The amount of drug administered intravenously is 400 ml twice daily at a rate of 40-60 drops per minute.
The tables below (Table 2-5) present the dynamics of MM and MDA in blood and urine before intravenous infusion of 400 ml of Rheosorbilact and in 20, 120 and 240 minutes upon completion of the infusion.
Table 3 demonstrates that after intravenous infusion of Rheosorbilact at a dose of 10-15 ml/kg of body weight in patients with purulent-septic diseases, an increase in MM and MDA concentrations in the blood plasma was seen in 20 minutes after the completion of infusion; then, MM and MDA concentrations in blood plasma decreased by an average of 22% from the initial level. In patients with pneumonia, isolated CCI, chronic cerebral circulatory insufficiency (Tables 2, 4, 5), by the 20th minute, a gradual reduction in the concentration of toxins was observed. In all studied groups, in 4 hours after Rheosorbilact infusion, MM and MDA concentrations in the blood plasma decreased by an average of 65–70%.
This phenomenon can be explained by the pharmacodynamic properties of Rheosorbilact. By increasing the plasma volume, decreasing the blood viscosity, Rheosorbilact provokes activation of microhemocirculation, dissolution of aggregates of blood corpuscles, restoration of exchange between blood and interstitium in all tissues of the body, which naturally leads to the washing out of MM and MDA from the microhemocirculatory bed into the central bloodstream.
In a parallel study of MM and MDA in urine after Rheosorbilact infusion, in 6 hours, MM and MDA concentration reached a maximum, which indicated an improvement in the rheological properties of blood caused by Rheosorbilact infusion and an increase in glomerular filtration. This fact is confirmed by an increase in MM and MDA urine concentrations (Table 6-9).
Table 2. Dynamics of MM and MDA indices in blood with infusion of Rheosorbilact in patients with pneumonias, M ± m

Note: here and in Tables 3–9: differences relative to data before administration are significant (* — P < 0.05; ** — P < 0.01; *** — P < 0.001); ^ — differences relative to data in 20 min after administration are significant (^ — P < 0.05; ^^ — P < 0.01; ^^^ — P < 0.001); ° — differences relative to data in 120 min after administration are significant (P < 0.001).
Table 3. Dynamics of MM and MDA indices in blood with infusion of Rheosorbilact in patients with purulent-septic diseases, M ± m

Table 4. Dynamics of MM and MDA indices in blood with infusion of Rheosorbilact in patients with isolated CCI, M ± m

Table 5. Dynamics of MM and MDA indices in blood with infusion of Rheosorbilact in patients with ICA reconstruction, M ± m

Table 6. Dynamics of MM and MDA indices in urine in 6 and 12 hours after infusion of Rheosorbilact in patients with pneumonias, M ± m
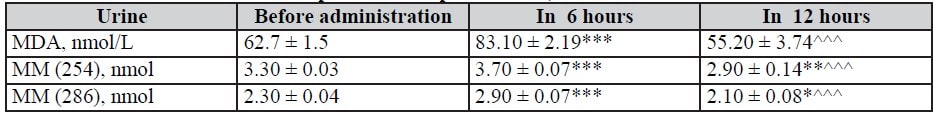
Table 7. Dynamics of MM and MDA indices in urine in 6 and 12 hours after infusion of Rheosorbilact in patients with isolated CCI, M ± m
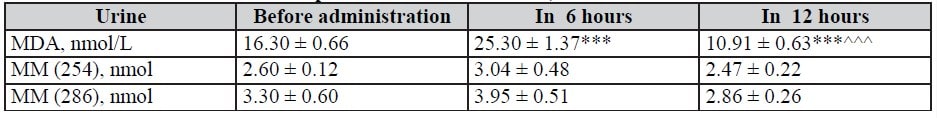
Table 8. MM and MDA indices in urine in 6 and 12 hours after infusion of Rheosorbilact in patients with ICA reconstruction, M ± m
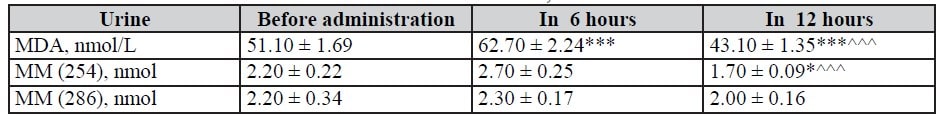
Table 9. MM and MDA indices in urine in 6 and 12 hours after infusion of Rheosorbilact in patients with purulent-septic diseases, M ± m
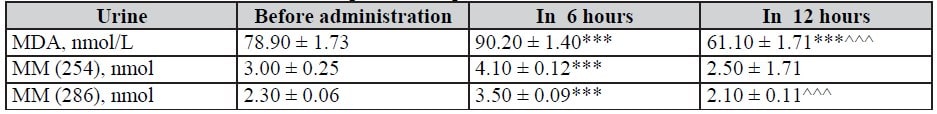
Table 10. Effects of Rheosorbilact on coagulation system, M ± m
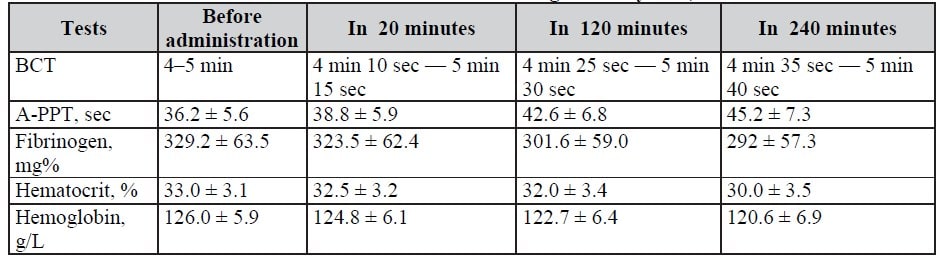
Notes: BCT – blood coagulation time; A-PPT – activated partial thromboplastin time; * – differences relative to data before administration are insignificant (P>0.05); ^ – differences relative to data in 20 minutes after administration are insignificant (P > 0.05).
Conclusions
- After the infusion of Rheosorbilact at a dose of 10-15 ml/kg, a significant reduction in the levels of middle molecules and malondialdehyde is observed. This reduction is associated with the effect of hemodilution and increased excretion by the kidneys. In 4 hours after the infusion of Rheosorbilact, in patients with various diseases, a reduction in middle molecules by 14-40%, malondialdehyde – by 36-72% from the initial value was observed.
- Rheosorbilact exerts a pronounced detoxifying effect and can be recommended as a basic drug for reducing intoxication in case of purulent-septic diseases, isolated CCI, pneumonias, in the postoperative period after reconstruction of the ICA.
- Administration of Rheosorbilact was associated with rapid normalization of blood coagulation indices, which indicated an improvement in blood rheology.
- Considering the polyfunctionality of Rheosorbilact, namely its hemodynamic action, pronounced detoxification effect, improvement in microcirculation and rheology, correction of fluid-and-electrolyte and acid-base balance, this drug product is recommended as a versatile solution in various surgical pathologies.
Authors:
Avakov V.Ye., Sayipov R.M., Davydov D.V., Begmatov S.Zh., Bazarov A.N., Tashkent Medical Academy, Department of Anesthesiology and Intensive Care, Republic of Uzbekistan
Literature:
- Baryshev, A.A. Gemodinamicheskiie plazmozameniteli [Hemodynamic plasma substitutes], Mir Meditsiny. – 2001. – No. 3-4. – P. 21-23 [in Russian].
- Gavrysiuk, V.K., Gumenyuk, N.I. Vliianiie reosorbilakta i reopoligliukina na sostoianiie gemokontsentratsii, viazkosti krovi i agregatsionnoi sposobnosti trombotsitov u bolnykh s hronicheskim legochnym serdtsem [Influence of rheosorbilact and rheopolyglucin on the state of hemoconcentration, blood viscosity and platelet aggregation ability in patients with chronic cor pulmonale]. Krovoobih ta gemostaz. – 2004. – No. 2. – P. 39-40 [in Ukrainian].
- Gabrielyan, N.I., Levitsky, E.R., Dmitriev, A.A. et al. Skriningovyi metod opredeleniia srednikh molekul v biologicheskih zhidkostiah [Screening method for the determination of middle molecules in biological fluids]. Recommended practices. – M., 1985. – 17 p. [in Russian].
- Gavrisyuk, V.K., Yachnik, A.I. Khronicheskoie legochnoie serdtse [Chronic cor pulmonale]. – K., 1997. – 96 p [in Russian].
- Gumenyuk, N.I., Lishnevskaya, V.Yu. vliianiie reosorbilakta na reologicheskiie svojstva krovi u bolnyh с ishemicheskoj bolezniu serdtsa I khronicheskim obstruktivnym bronhitom [The effect of rheosorbilact on the rheological properties of blood in patients with ischemic disease and chronic obstructive bronchitis]. Ukrainskij pulmunologicheskii zhurnal. – 2003. – No. 3. – P. 38-40 [in Russian].
- Molchanov, I.V., Kosachenko, V.M., Gorbachevsky, Yu.V. Sovremennye metody primeneniia kolloidnykh rastvorov v infusionno-transfusionnoj terapii [Modern methods for the use of colloidal solutions in infusion-transfusion therapy]. Vestnik sluzhby krovi. – 1999. – P. 35-40 [in Russian].
- Kogonia, M.Z., Pasternak, G.I., Tkacheva, M.Yu. Opyt primeneniia otechestvennykh preparatov reosorbilakt i sorbilakt v kompleksnom lechenii bolnykh s ostrym pankreatitom v usloviiah otdeleniia intensivnoj terapii [Experience of using domestic drugs rheosorbilact and sorbilact in the combined treatment of patients with acute pancreatitis in intensive care units]. Ukrainskij khimioterapevticheskij zhurnal. – 2008. – No. 1–2 (22). – P. 172-175 [in Russian].
- Dintenfass L., Julian D.G., Miller G.E. Viscosity of blood in normal subjects and in patients suffering from coronary occlusion and arterial thrombosis // Am. Heart J. — 1986. — Vol. 111. — P. 587600.
- Dormandy J.A., Edelman J.B. High blood viscosity an aetiological factor in venous thrombosis // Brit. J. Surg. — 1973. — Vol. 60, № 3. — P. 187-190.
- Dormandy J.A. Clinical evidence of link between haemorheology and thrombosis//Hemostasis. — 1982. — Vol. 12, № 1. — P. 168-170.
- Haljamäe H., Dahlqvist M., Walentin F. Artificial colloid in clinical practice: pros and cons // Clin. Anaesthesiol. — 1997. — Vol. 1. — P. 49-79.
- Simone G., Devereux R.B., Chien S. et al. Relation of blood viscosity to demographic and physiologic variables and to cardiovascular risk factors in apparently normal adults // Circulation. — 1990. — Vol. 81, № 1. — P. 107-117.





