Neurological disorders in patients who had COVID-19: how to treat during a pandemic
The article provides an overview of the world literature data on lesions of the nervous system in patients who had COVID-19. The results of an open comparative study on the effectiveness of combined use of Xavron, Tivor-L and Xylat in patients with neurological disorders in the post-COVID period are also presented. The positive effect of the above combination of drugs on cognitive functions was demonstrated, which manifested itself in increasing the rate of mental activity, reducing the manifestations of the severity of asthenia, improved the indicators of the quality of life.
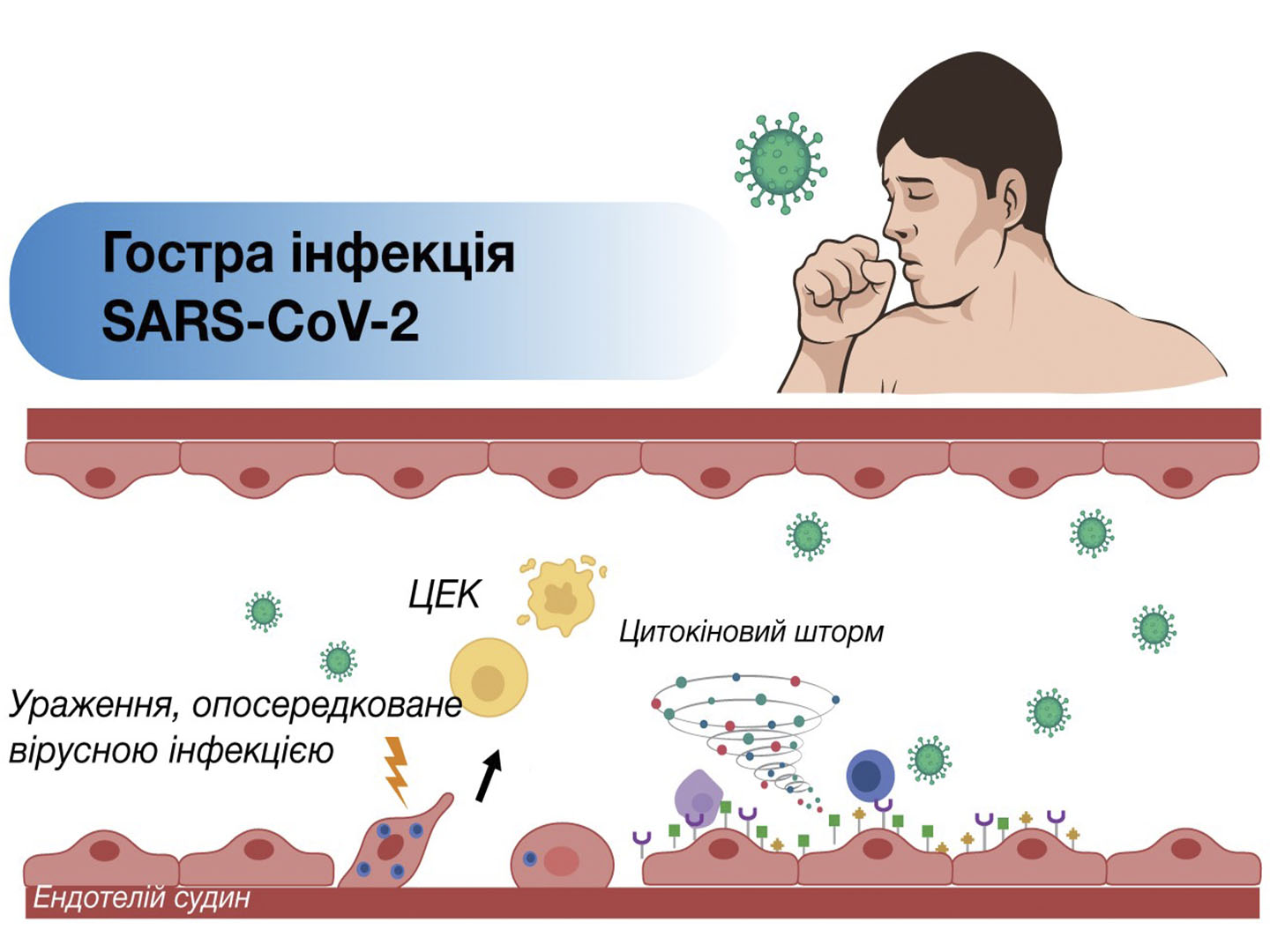
In post-COVID patients with neurological complications, serum levels of the pro-inflammatory marker cytokines IL-6 and VEGF were elevated. The addition of Xavron, Tivorel and Xylat to the basic therapy led to a decrease in the levels of IL-6 and VEGF, which suggests an anti-inflammatory effect of the proposed treatment regimen. The efficacy and safety of the syndromic-pathogenetic complex determines the prospects for its appointment for the treatment of patients with neurological post-COVID disorders.
Keywords: COVID-19 pandemic, neurological disorders, treatment, post-COVID.
Authors:
T.S. Mishchenko, V.M. Mishchenko V.V. Sokolik, A.V. Fisenko, I.V. Zdesenko V.N.
Karazin Kharkiv National University, Kharkiv, Ukraine
State Institution “Institute of Neurology, Psychiatry and Narcology of the National Academy of Medical Sciences of Ukraine”, Kharkiv, Ukraine
Literature:
- Niu , Shen L., Huang B. et al. Non-invasive bioluminescence imaging of HCoV-OC43 infection and therapy in the central nervous system of live mice. Antiviral. Res. 2020. 173. 104646.
- Lodigiani , Iapichino G., Carenzo L. et al. Venous and arterial thromboembolic complications in COVID-19 patients admitted to an academic hospital in Milan, Italy. Thromb. Res. 2020. 191. 9-14.
- Hoffmann , Kleine-Weber H., Schroeder S., Krüger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.H., Nitsche A., Müller M.A., Drosten C., Pöhlmann S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020. 181(2). 271-280.e8.
- https://doi.org/10.1016/j.cell.2020.02.052.
- Rothan A., Byrareddy S.N. The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J. Autoimmun. 2020. 109. 102433.
- Filatov , Sharma P., Hindi F. et al. Neurological complications of coronavirus disease (COVID-19): Encephalopathy. Cureus. 2020. 12(3). e7352.
- Ye , Ren Y., Lv T. Encephalitis as a clinical manifestation of COVID-19. Brain Behav. Immun. 2020. S0889-1591(20)30465-7.
- Moriguchi , Harii N., Goto J. et al. A first case of meningitis/ encephalitis associated with SARS-Coronavirus-2. Int. J. Infect. Dis. 2020. 94. 55-58.
- Duong , Xu P., Liu A. Meningoencephalitis without respiratory failure in a young female patient with COVID-19 infection in Downtown Los Angeles, early April 2020. Brain Behav. Immun. 2020. 87. 33.
- Zhao , Huang J., Dai D. et al. Acute myelitis after SARS-CoV-2 infection: a case report. MedRxiv. 2020.
- Wu , Xu X., Chen Z., Duan J., Hashimoto K., Yang L., Liu C., Yang C. Nervous system involvement after infection with COVID-19 and other coronaviruses. Brain Behav. Immun. 2020. 87. 18-22. doi: 10.1016/j.bbi.2020.03.031.
- Padroni , Mastrangelo V., Asioli G.M. et al. Guillain-Barré syndrome following COVID-19: new infection, old complication? [published online ahead of print, 2020 Apr 24]. J. Neurol. 2020. 1-3.
- Virani , Rabold E., Hanson T. et al. Guillain-Barré syndrome associated with SARS-CoV-2 infection [published online ahead of print, 2020 Apr 18]. IDCases. 2020. 20. e00771.
- Camdessanche P., Morel J., Pozzetto B. et al. COVID-19 may induce Guillain-Barré syndrome. Rev. Neurol. (Paris). 2020. 176(6). 516-518.
- Sedaghat , Karimi N. Guillain Barre syndrome associated with COVID-19 infection: A case report. J. Clin Neurosci. 2020. 76. 233-235.
- Nguyen-Huynh N., Tang X.N., Vinson D.R. et al. Acute Stroke Presentation, Care, and Outcomes in Community Hospitals in Northern California During the COVID-19 Pandemic. Stroke (IF 7.190). Pub Date: 2020-08-07. DOI: 10.1161/ strokeaha.120.031099.
- Li Y. Acute cerebrovascular disease following COVID-19: a single center, retrospective, observational study. SSRN J. 2020.
- Mao , Jin H., Wang M., Hu Y., Chen S., He Q., Chang J., Hong C., Zhou Y., Wang D., Miao X., Li Y., Hu B. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wu- han, China. JAMA Neurol. 2020. e201127. https://doi.org/10.1001/ jamaneurol.2020.1127.
- Міщенко Т.С., Міщенко В.М. Неврологічні ускладнення у пацієнтів з COVID-19. Міжнародний науково-практичний журнал «Психіатрія, неврологія та медична психологія» ХНУ ім. В.Н. Каразіна. № 16. С. 23-33.
- Espinosa P.S., Rizvi , Sharma P., Hindi F., Filatov A. Neurological Complications of Coronavirus Disease (COVID-19): Encephalopathy, MRI Brain and Cerebrospinal Fluid Findings: Case 2. Cureus. 2020. 12. e7930.
- Tay Z., Poh C.M., Rénia L., MacAry P.A., Ng L.F.P. The trinity of COVID-19: immunity, inflammation and intervention. Nat. Rev. Immunol. 2020. 20. 363-74. doi:10.1038/s41577-020-0311-8 pmid: 32346093. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7187672/
- Barker-DaviesM., O’Sullivan O., Senaratne K.P. et al. The Stanford Hall consensus statement for post-COVID-19 rehabilitation. Br. J. Sports Med. 2020. 54. 949-959.
- Gemelli Against COVID-19 Post-Acute Care Study Post-COVID-19 global health strategies: the need for an interdisciplinary approach. Aging Clinical and Experimental Research. https:// doi.org/10.1007/s40520-020-01616-x.
- Reznik E. et al. Edaravone: A potential treatment for the COVID-19-induced induced inflammatory syndrome? Pharmacol. Res. 2020 Jun 30. 160. 105055.
- Johns Hopkins Coronavirus Resource Center. Accessed at https://coronavirus.jhu.edu/map.html.
- Chu , Chan J.F., Yuen T.T. et al. Comparative tropism, replication kinetics, and cell damage profiling of SARS-CoV-2 and SARS-CoV with implications for clinical manifestations, transmissibility, and laboratory studies of COVID-19: an observational study. Lancet Microbe. 2020. 1(1). 14-23.
- Hashimoto , Sanui M., Egi M. et al. The clinical practice guideline for the management of ARDS in Japan. J. Intensive Care. 2017. 5. 50.
- Saito , Fujii Y., Yashiro M. et al. Inhibitory Effects of Edaravone, a Free Radical Scavenger, on Cytokineinduced Hyperpermeability of Human Pulmonary Microvascular Endothelial Cells: A Comparison with Dexamethasone and Nitric Oxide Synthase Inhibitor. Acta Med. Okayama. 2015. 69(5). 279-290.
- Kanjanaumporn, Aeumjaturapat S., Snidvongs K., Seresirikachorn K., Chusakul S. Smell and taste dysfunction in patients with SARSCoV-2 infection: A review of epidemiology, pathogenesis, prognosis, and treatment options. Asian Pac. J. Allergy Immunol. 2020. 38. 69-77.
- Cantuti-Castelvetri , Ojha R. et al. Neuropilin-1 facilitates SARS-CoV-2 cell entry and infectivity. Science. 2020. 370. 856-860.
- Adamczyk-Sowa , Niedziela N., Kubicka-Baczyk K., Wier- zbicki K., Jaroszewicz J., Sowa P. Neurological symptoms as a clinical manifestation of COVID-19: Implications for internists. Polish Arch. Intern. Med. 2020.
- Alquisiras-Burgos , Peralta-Arrieta I., Alonso-Palomares L.A. et al. Neurological Complications Associated with the Blood-Brain Barrier Damage Induced by the Inflammatory Response During SARS-CoV-2 Infection. Mol. Neurobiol. 2021. 58. 520-535. https://doi.org/10.1007/s12035-020-02134-7.
- Al-Sarraj S., Troakes C., Hanley B., Osborn M., Richard- son P., Hotopf M., Bullmore E., Everall I.P. Invited Review: The spectrum of neuropathology in COVID-19. Neuropathol. Appl. Neurobiol. 2021 Feb. 47(1). 3-16. doi: 10.1111/nan.12667. Epub 2020 Oct PMID: 32935873.
- Poyiadji , Shahin G., Noujaim D., Stone M., Patel S., Griffith B. COVID-19-associated acute hemorrhagic necrotizing encephalopathy: CT and MRI features. Radiology. 2020. 201187.
- Дзяк Л.А., Цукаленко О.С., Чеха К.В., Сук В.М. Сучасні аспекти нейрогенезу й неврологічних проявів COVID-19. Міжнародний неврологічний журнал. 2021. Т. 17. № 2. С. 6. DOI:https://org/10.22141/22240713.17.2.2021.229887.
- Зозуля І.С., Мардзвік В.М., Мардзвік М.В. Судинні неврологічні ускладнення в пацієнтів з COVID-19. Український медичний часопис. ІІ/IV. 2(142). DOI: 10.32471/umj.1680- 3051.142.204731.
- Копчак О.О. Особливості когнітивних порушень при COVID-19. Міжнародний неврологічний журнал. 2021. Т.№3.С.12.DOI:http://dx.doi.org/https://doi.org/10.22141/2224-0713.17.3.2021.231569.
- Oliveira et al. Neuro-COVID frequency and short-term outcome in the northern Portuguese population. European Journal of Neurology. https://doi.org/10.1111/ene.14874.
- Fartushna Yе., Palahuta H.V., Yevtushenko S.K. Neurological and neuropsychiatric manifestations and complications of SARS-CoV-2 infection: a narrative review and a case presentation in a previously healthy young white adult. Міжнародний неврологічний журнал. 2021. Т. 17. № 1. С. 36. DOI: https://doi. org/10.22141/2224-0713.17.1.2021.226914.
- Panariello , Cellini L., Speciani M., De Ronchi D., Atti A.R. How Does SARS-CoV-2 Affect the Central Nervous System? A Working Hypothesis. Front Psychiatry. 2020. 11. 582345. Published 2020 Nov 16. doi: 10.3389/fpsyt.2020.582345.
- Zhou , Zhang M., Wang J. et al. Sars-CoV-2: Underestimated damage to nervous system [published online ahead of print, 2020 Mar 24]. Travel Med. Infect. Dis. 2020. 101642.
- Pilotto , Odolini S., Masciocchi S. et al. Steroidresponsive encephalitis in coronavirus disease 2019 [published online ahead of print, 2020 May 17]. Ann. Neurol. 2020. 10.1002/ana.25783.
- Lin Y., Lee K.Y., Ro L.S., Lo Y.S., Huang C.C.,Chang K.H. Clinical and cytokine profile of adult acute necrotizing encephalopathy. Biomed J. 2019. 42(3). 178-186. https://doi. org/10.1016/j.bj.2019.01.008.
- Klok A., Kruip M., van der Meer N.J.M., Arbous M.S., Gommers D., Kant K.M. et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb. Res. 2020. 191. 145-7.
- Asadi-Pooya A., Simani L. Central nervous system manifestations of COVID-19. A systematic review. J. Neurol. Sci. 2020. 413. 116832.
- Carfì , Bernabei R., Landi F., Group ftGAC-P-ACS. Persistent Symptoms in Patients After Acute COVID-19. J. Am. Med. Assoc. 2020. 324(6). 603-605. DOI: 10.1001/jama.2020.12603.
- Armocida , Pesce A., Raponi I., Pugliese F., Valentini V., Santoro A., Berra L.V. Anosmia in COVID-19: severe acute respiratory syndrome coronavirus 2 through the nasoliary epithelium and a possible spreading way to the central nervous system — A purpose to study. Neurosurgery. 2020. https://doi.org/10.1093/neuros/nyaa204.
- Mahalaxmi , Kaavya J., Mohana Devi S., Balachandar V. COVID-19 and olfactory dysfunction: A possible associative approach towards neurodegenerative diseases. J. Cell. Physiol. 2021. 236. 763-770. https://doi.org/10.1002/jcp.29937.
- Parry H., Wani A.H., Yaseen M. Neurological Dysfunction in Coronavirus Disease-19 (COVID-19). Acad. Radiol. 2020. 27. 1329-1330.
- Huang , Wang Y., Li X. et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020. 395. 497-506.
- Helms , Kremer S., Merdji H. et al. Neurologic features in severe SARS-CoV-2 infection. N. Engl. J. Med. 2020. 382(23). 2268- 2270.
- Тріщинська М.А., Кононов О.Є., Бельська І.В. Патогенетично обгрунтована профілактика цереброваскулярних захворювань у хворих із коронавірусною інфекцією. Міжнародний неврологічний журнал. Т. 16. № 7. С. 23-31.
- Лунева И.Е., Полищук Р.В., Чернобаева Л.С., Бормин А.А., Антипят Н.А., Рябинкина Ю.В., Пирадов М.А. Острый некротический энцефалит, ассоциированный с вирусом гриппа, у взрослых. Журнал неврологии и психиатрии им. С.С. Корсакова. 2020. 120(4). 101-105. https://doi.org/10.17116/jnevro2019119121100.
- Wang L., Iwanami J., Min L.J., Tsukuda K., Nakaoka H., Bai H.Y., Shan B.S., Kan-No H., Kukida M., Chisaka T., Ya- mauchi T., Higaki A., Mogi M., Horiuchi M. Deficiency of angio- tensin-converting enzyme 2 causes deterioration of cognitive func- tion. NPJ Aging Mech. Dis. 2016. 2. 16024. https://doi.org/10.1038/ npjamd.2016.24.
- Ayoubkhani , Khunti K., Nafilyan V., Maddox T., Humberstone B., Diamond I. et al. Post-COVID syndrome in individuals admitted to hospital with COVID-19: retrospective cohort study. BMJ. 2021. 372. n693. doi: 10.1136/bmj.n693
- Greenhalgh , Knight M., A’Court C., Buxton M., Husain L. Management of post-acute covid-19 in primary care. Br. Med. J. 2020. 370. m3026. DOI: 10.1136/bmj.m3026.
- СOVID-19 rapid guideline: managing the long-term effects of COVID-19. NICE guideline [NG188]. Published date: 18 December 2020. https://www.nice.org.uk/guidance/ng188/chapter/5-Management.
- Lopez-Leon , Wegman-Ostrosky T., Perelman С., Sepulveda R., Rebolledo P.A., Cuapio А., Villapol S. More than 50 Longterm effects of COVID-19: a systematic review and meta-analysis. Doi: https://doi.org/10.1101/2021.01.27.21250617.
- Sharma , Lioutas V.-A., Madsen T., Clark J., O’Sullivan J., Elkind M.S.V., Willey J.Z., Marshall R.S., Selim M.H., Greer D., Tirschwell D.L., Burton T., Boehme A. Decline in stroke alerts and hospitalisations during the COVID-19 pandemic. Stroke Vasc Neurol. 2020 Dec. 5(4). 403-405. Article in English | MEDLINE | ID: covidwho-1318203.
- Guedj , Campion J.Y., Dudouet P. 18F-FDG brain PET hypometabolism in patients with long COVID. Eur. J. Nucl. Med. Mol. Imaging. 2021. https://doi.org/10.1007/s00259-021-05215-4.
- Carvalho-Schneider , Laurent E., Lemaignen A., Beaufils E., Bourbao-Tournois C., Laribi S. et al. Follow-up of adults with noncritical COVID-19 two months after symptom onset. Clin. Microbiol. Infect. 2020. DOI: 10.1016/j.cmi.2020.09.052.
- Spinato , Fabbris C., Polesel J. et al. Alterations in Smell or Taste in Mildly Symptomatic Outpatients with SARS-CoV-2 Infection. JAMA. 2020. 323(20). 2089-2090.
- Noor-Ul-Huda , Ain S., Salman Y. The Effects of COVID-19 on Hypothalamus: Is it Another Face of SARS-CoV-2 That May Potentially Control the Level of COVID-19 Severity? DOI: 10.13140/RG.2.2.24482.25289.
- Goërtz M.J., Van Herck M., Delbressine J.M., Vaes A.W., Meys R., Machado F.V.C. et al. Persistent symptoms 3 months after a SARS-CoV-2 infection: the post-COVID-19 syndrome? ERJ Open Res. 2020. DOI: 10.1183/23120541.00542-2020.
- Голубовська О.А. Постковідний синдром: патогенез та основні напрями реабілітації. Здоров’я України 21 сторіччя.№ 3(496).
- Paterson W., Brown R.L., Manji H., Zandi M.S. Contrib- uted equally to this work. Brain. 2020. Vol. 143. Issue 10. https://doi. org/10.1093/brain/awaa240.
- Weir К. COVID-19 attacks the brain. Monitor on Psychology. Vol. 51. № 8. Р. 20.
- Branca М. Early details of brain damage in COVID-19 patients. The Harvard Gazette. November 18, 2020.
- Baird S., Montine T.J. Multiplex immunoassay analysis of cytokines in idiopathic inflammatory myopathy. Arch. Pathol. Lab. Med. 2008. 132(2). 232238.
- Varga , Flammer A.J., Steiger P. et al. Endothelial cell infection and endotheliitis in COVID-19. The Lancet. 2020. 395(2). 1417-1418.
- Desforges , Le Coupanec A., Stodola J.K. Human coronaviruses: viral and cellular factors involved in neuroinvasiveness and neuropathogenesis. Virus Res. 2014 Dec 19. 194. 145-58.
- Najjar , Najjar A., Chong D.J. et al. Central nervous system complications associated with SARS-CoV-2 infection: integrative concepts of pathophysiology and case reports. Journal of Neuroinflammation. 2020. 17. 231.
- Zhang , Zhao Y., Zhang F. The use of anti-inflammatory drugs in the treatment of people with severe coronavirus disease 2019 (COVID-19): The Perspectives of clinical immunologists from China. Clinical Immunology. 2020. 214. 108393.
- Yang Y. Edaravone Alleviated Propofol-Induced Neurotoxicity in Developing Hippocampus by mBDNF/TrkB/PI3K Pathway. Drug Devel. Ther. 2021. 15. 1409-1422.
- Негрич Т.І. Застосування препаратів едаравону, цитиколіну й електролітів та L-аргініну в пацієнтів із гострим порушенням мозкового кровообігу. НейроNEWS. 2020. 9(120). 12-19.
- Watanabe et al. How is edaravone effective against acute ischemic stroke and amyotrophic lateral sclerosis? J. Clin. Biochem. Nutr. 2018 Jan. 62(1). 20-38
- Znong H. Efficacy and safety of current therapeutic options for COVID-19lessons to be learnt from SARS and MERS epidemic: A systematic review and meta-analysis. Pharmacol Res. 2020 Jun. 157.
- Загайко А.Л., Брюханова Т.О., Шинкарьов А.А., Мельник Н.В. Метаболічні ефекти карнітину, роль у розвитку патологій та перспективи клінічного застосування (огляд). Український біофармацевтичний журнал. № 6.
- Smeis M., Garssen B.J., Bonke B., DeHaes J.C. Піе multidimensional fatigue inyentory (MFI) psychometric qualities of an instrument to assess fatigue. Journal of Psychosomatic Research. 1995. 39. 315-325.
- Ханин Ю.Л. Краткое руководство к применению шкалы реактивной личностной тревожности Ч.Д. Спилбергера. Л.: ЛНИИФК, 1976.
- Nasreddine S., Phillips N.A., Bédirian V., Charbonneau S., Whitehead V., Collin I., Cummings J.L., Chertkow H. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J. Am. Geriatr. Soc. 2005. 53(4). 695-699. https://doi. org/10.1111/j.1532-5415.2005.53221.x.
- Brazier , Roberts J., Deverill M. The estimation of a preference-based measure of health from the SF-36. J. Health Econ. 2002. 21(2). 271-292. https://doi.org/10.1016/s0167-6296(01)00130-8.