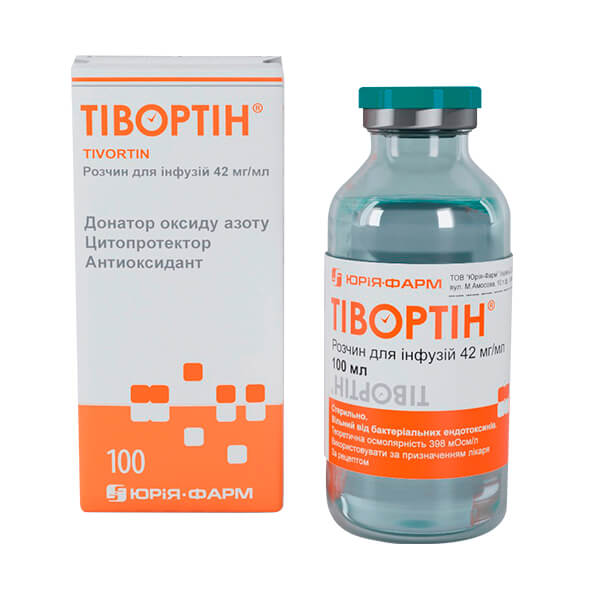
Tivortin®
Active substance formulated into Tivortin® – L-arginine amino acid — is the only substrate for nitric oxide (NO) synthesis in humans.
Tivortin® is a potent vasodilator, vascular tone regulator.
Tivortin® is a universal angioprotector:
♦ reduces activation and adhesion of leukocytes to the endothelium;
♦ has antithrombotic properties, reduces platelets adhesion, their activation and aggregation;
♦ prevents pathological vascular remodeling and atherosclerosis progression.
Forms of release in Georgia:
- 42 mg/ml, 100 ml in glass bottle.
Xylat-Tivortin course Life Extension
40+ is a wonderful age to discover yourself, to accept your body as the greatest value, but this age is special.
During this period of life, a woman needs additional care: energy saturation, acceleration of metabolism, improvement of microcirculation of tissues and organs to normalize their work and prevent the development of cardiovascular diseases.
Xylat-Tivortin course is designed specifically to improve the quality of life of women.
Xylat-Tivortin course Life Extension:
- provides energy and emotional uplift
- raises the standard of living and improves the women status at menopausal age
- reduces the manifestations of menopause
- edema decreases, due to which body weight decreases
The mechanism of influence of the Life Extension course on a woman’s body:
- restoration of energy metabolism in cells
- improving carbohydrate metabolism
- support of vascular endothelial function
- improvement of microcirculation

Watch the video report (in Ukrainian) of Professor Lakhno Igor Viktorovich about Xylato-Tivortin Life Extension course (from 18:35 minute):
See the instructions for medicines included in the Xylat-Tivortin course:
Xylat || Tivortin || Tivortin aspartate
How supplied
Sales markets
Azerbaijan, Georgia, Kazakhstan, Kyrgyzstan, Moldova, Tajikistan, Turkmenistan, Uzbekistan, Ukraine.
Instruction
PACKAGE LEAFLET:
INFORMATION FOR THE USER
TIVORTIN
Solution for infusion
Read all of this leaflet carefully before you start using this medicine because it contains important information for you.
- Keep this leaflet. You may need to read it again.
- If you have any further questions, ask your doctor or pharmacist.
- If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet.
What is In this leaflet:
- What TIVORTIN is and what it is used for
- What you need to know before you use TIVORTIN
- How to use TIVORTIN
- Possible side effects
- How to store TIVORTIN
- Contents of the pack and other information
1. What TIVORTIN is and what it is used for
TIVORTIN is an solution for infusion given into your blood by a drip (intravenous infusion). The product contains amino acid (component used to build proteins) in a bottle. He is converted in the body into a chemical called nitric oxide. Nitric oxide causes blood vessels to open wider for improved blood flow. L-arginine is an essential amino acid that may play an important role in the treatment of heart disease, pulmonary diseases, hepatitis, including after inflectional diseases and operative treatment and other. L-arginine also lowers cholesterol, stimulates the release of growth hormone, insulin, and other substances in the body.
2. What you need to know before you use TIVORTIN
Do not use TIVORTIN:
- if you are allergic (hypersensitive) to the ingredients of this medicine (listed in section 6)
- if your body has problems using amino acids
- if you have a serious kidney disease
- if you have too much sugar in your blood (hyperglycaemia) which is uncontrolled
- if you are in an unstable condition, such as metabolic acidosis (a disturbance resulting in too much acid in the blood).
Warnings and precautions
Talk to your doctor before using TIVORTIN if you have:
- kidney problems
- pancreatitis (inflammation of the pancreas)
- diabetes mellitus
If you during the infusion get fever, rash, swelling, difficulty in breathing, chills, sweating, nausea or vomiting, tell the health care professional immediately because these symptoms might be caused by an allergic reaction.
Your doctor may regularly need to check your blood for kidney function tests and other values.
TIVORTIN is not meant for newborn babies or children younger than 3 years old.
Children and adolescent
TIVORTIN is not meant for newborn babies or children younger than 3 years old.
Other medicines and TIVORTIN
Tell your doctor if you are taking, have recently taken or might take any other medicines, even without prescription.
Pregnancy and breast-feeding
Data from using TIVORTIN during pregnancy or breastfeeding is lacking. TIVORTIN should therefore be given to pregnant or breast-feeding women only if the doctor find it necessary. The use of TIVORTIN may be considered during pregnancy and breastfeeding, as advised by your doctor
Driving and using machines
Caution must be exercised when driving or operating machinery as the drug can cause dizziness.
3. How to use TIVORTIN
Always use this medicine exactly as your doctor has told you. Check with your doctor if you are not sure.
Your doctor will decide on the dose for you individually depending on your body weight and function. TIVORTIN will be given to you by a health care professional.
If you use more TIVORTIN than you should
It is unlikely that you will receive too much medicine as TIVORTIN is given to you by a health care professional.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
- a raised body temperature.
- Inflammation in superficial peripheral veins in connection to the injection site.
- nausea
- dry mouth
- vomiting
- chills
- headache
- low or high blood pressure
- pain in the region of heart
- difficulty in breathing
- fast heart beat (tachycardia)
- hypersensitivity reactions (that can give symptoms like swelling, fever, fall in blood pressure, skin rashes, wheals (raised red areas), flushing, headache)
- sensations of hot
- pain in the neck, back, bones and breast
- paleness
- light blue coloured lips and skin (because of too little oxygen in the blood)
- dizziness
- apprehension
- asthenia
- convulsions
- tremor
If you get any side effects, talk to your doctor or pharmacist. This includes any side effects not listed in this leaflet.
5. How to store TIVORTIN
Keep this medicine out of the sight and reach of children. Store below 30 °C in original package. Do not use this medicine after the expiry date which is stated on the label on the bag and box. The expiry date refers to the last day of that month.
6. Further information
What TIVORTIN contains
The active substance is L-arginini hydrochloridum
1 mL contains 42 mg of arginine hydrochloride (100 mL contain 20 mmol of arginine and 20 mmol of chlorides);
auxiliary components: water for injections.
The following information is intended for healthcare professionals only:
Warnings and precautions for use
Since an increased risk of infection is associated with the use of any peripheral vein, strict aseptic precautions should be taken to avoid any contamination.
Serum glucose, electrolytes and osmolarity as well as fluid balance, acid-base status and kidney and enzyme tests should be monitored.
Any sign or symptom of anaphylactic reaction (such as fever, shivering, rash or dyspnoea) should lead to immediate interruption of the infusion.
During the use of Tivortin, it should be taken into consideration that the drug can cause a pronounced and refractory hyperkalemia and a concurrent renal failure in the patients who are taking or were taking spironolactone. The prior administration of potassium-sparing diuretics may increase the blood potassium level. The concurrent use of amynophyllin may increase the blood insulin level.
Thrombophlebitis may occur if peripheral veins are used for infusions. The catheter insertion site should be evaluated daily for local signs of thrombophlebitis.
Method of administration
Intravenous use, infusion into a peripheral or a central vein.
The drug is administered intravenously by drops with rate of 10 dpm during the first 10-15 min, then the rate can be increased up to 30 dpm.
The daily dose of the drug is 100 mL of solution.
In patients with severe circulation failure in central and peripheral vessels, in case of pronounced intoxication, hypoxia, asthenic states the dose can be increased up to 200 mL per day.
The maximal infusion rate should not exceed 20 mmol/hour.
The dose for children under 12 is 5-10 mL per 1 kg of body mass per day.
The dose for treatment of metabolic alkalosis is calculated as follows:
arginine hydrochloride (mmol)
______________________________________ x 0.3 kg of body mass
bases excess (Be) (mmol/L)
The administration is started with a half of the calculated dose. The additional correction can be made after the results of renewed acid-base balance have been obtained.
Noncompatibility
The drug is non-compatible with thiopental.
What TIVORTIN looks like and contents of the pack
Solution is transparent, colorless or slightly yellowish-brownish liquid.
Pack sizes:
100 ml in 1 bottle in a package.
Registrant
«Yuria-Pharm» LLC
10, M. Amosova street, Kyiv, Ukraine, 03680.
Tel.: +38 (044) 275-92-42.
E-mail: uf@uf.ua
Manufacturer
«Yuria-Pharm» LLC
108, Kobzarska street, Cherkassy, Cherkassy region, Ukraine, 18030.
Tel.: +38 (044) 281-01-01.
Local Technical Representative (LTR)
HARLEYS LTD-NAIROBI
Date of last revision:
28.11.2019
Articles
- Cardioprotector effect of nitrogen oxide donor in patients with hypertension
- State of metabolic processes and ways to improve them in premenopausal women due to the Life Extension Strategy
- Pathogenetic pancreoprotection in diabetic ketoacidosis
- Healthy nephrone — a guaranty of stable blood pressure
- Metabolic regulation in mitochondria as a prospective way of body rejuvenation
- What should a practitioner know about endothelial dysfunction?
- Pain syndrome in adenomyosis. finding new pathogenesis links and non-hormonal correction opportunities. Literature review (Ukrainian language).
- " Long-haulers ": patients with post-covid syndrome in routine clinical practice
- School of endocrinologist: some aspects of infusion treatment of diabetes
- L-arginine in norm and pathology
- The role of L-arginine in the treatment of cardiovascular diseases: the focus is on the choice of effective dose
- A safe and effective non-prohibited drug for stimulating the performance of athletes
- The efficacy of Tivortin® in the treatment of patients with stable effort angina
- A novel approach to the therapy of placental dysfunction using L-arginine
- Tivortin® in the therapy of placental dysfunction
- L-Arginine in coronary heart disease: the research continues
- L-arginine from the viewpoint of evidence-based medicine